Clinicubes on Twitter: "✔️ICH GCP E6 (R2) FDA Guidance has been released. ❗An important read for all clinical research professionals: ➡️https://t.co/ok3QmHsaWI #ClinicalTrials #GCP #GxP #Trals #FDA #clinicalresearch #ICHGCPE6 https://t.co ...

ICH GCP Guidelines E6 Revision, R2 Addendum – Changes Impacting Sponsors-CRO-Sites – Compliance Trainings

Introduction To Investigators Responsibilities With Good Clinical Practice | PDF | Institutional Review Board | Clinical Trial

Certified Copies and ALCOA-C: Essentials Post ICH GCP E6 (R2) Addendum - Life Science Training Institute
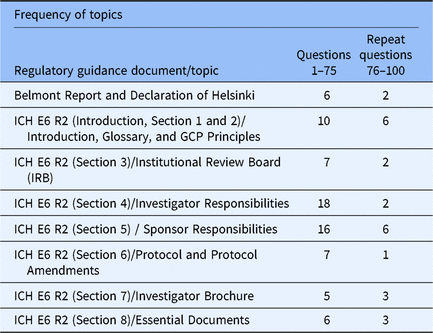
Creating and testing a GCP game in an asynchronous course environment: The game and future plans | Journal of Clinical and Translational Science | Cambridge Core

good clinical practice.pdf - E6(R2) Good Clinical Practice: Integrated Addendum to ICH E6(R1) Guidance for Industry U.S. Department of Health and Human | Course Hero

The Impact of ICH GCP E6 Guideline R2 Revisions on Sponsors, Sites, Contract Research Organizations and Vendors | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services


.jpg)