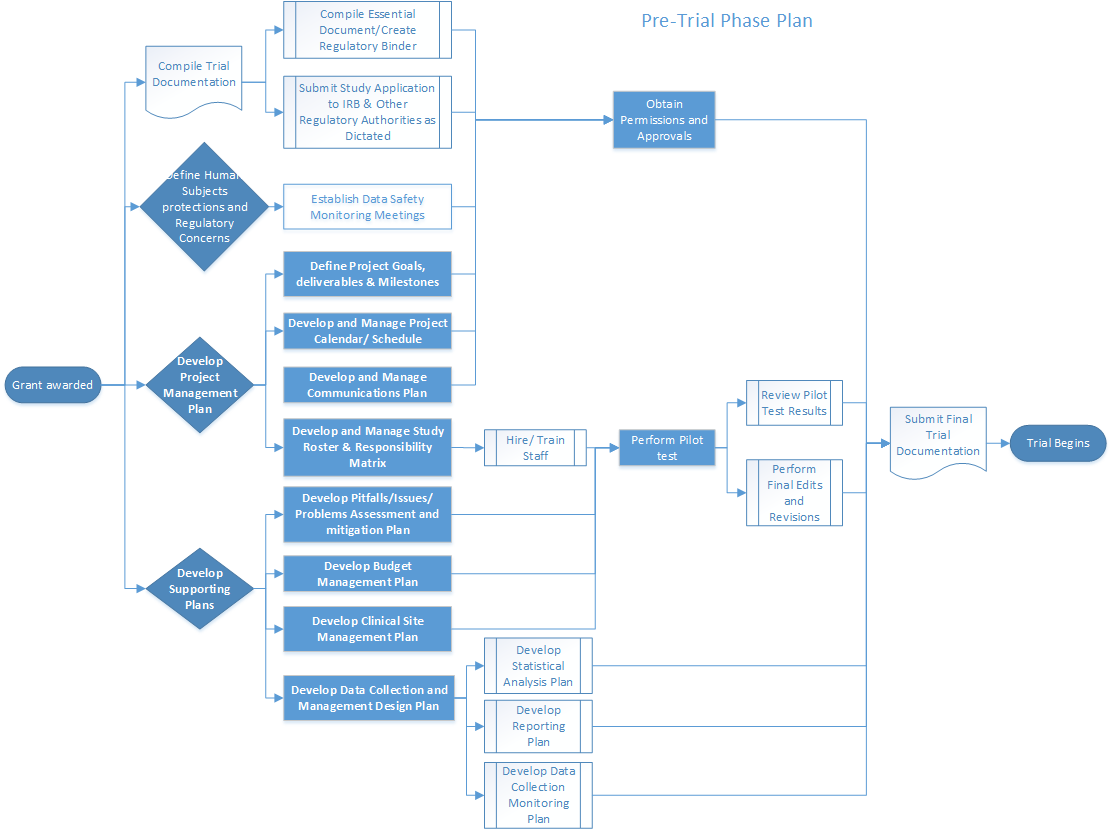
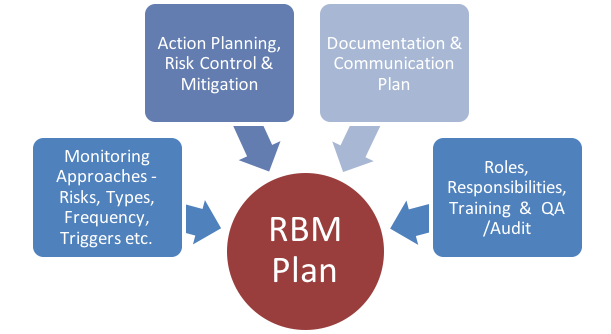
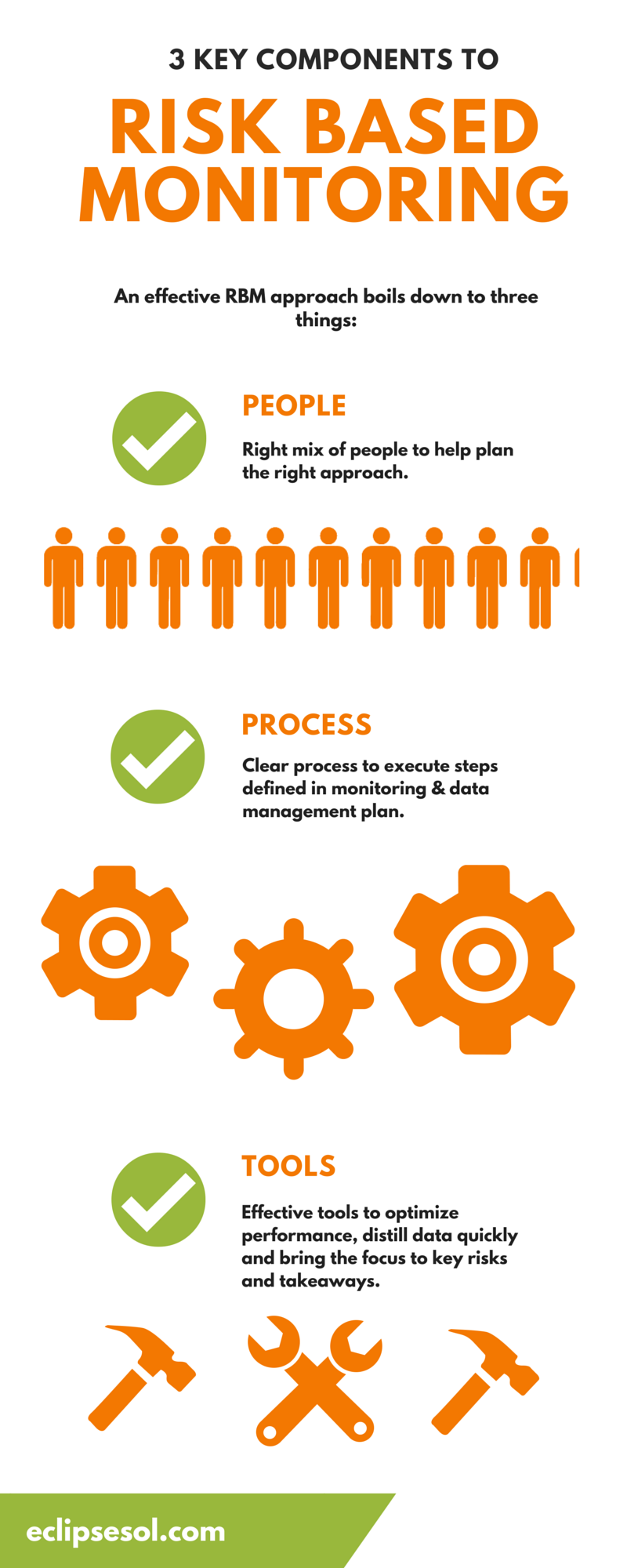
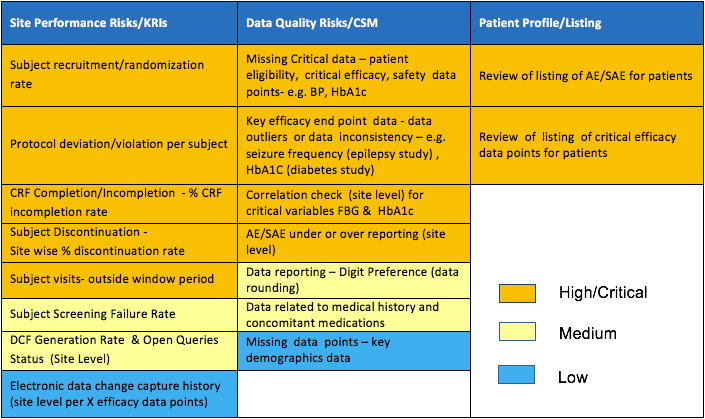
Validation of a risk-assessment scale and a risk-adapted monitoring plan for academic clinical research studies — The Pre-Optimon study - ScienceDirect
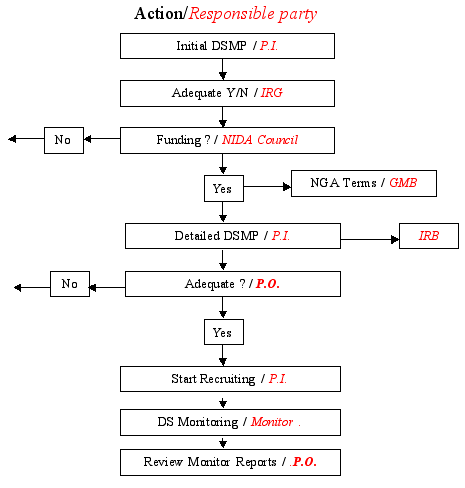
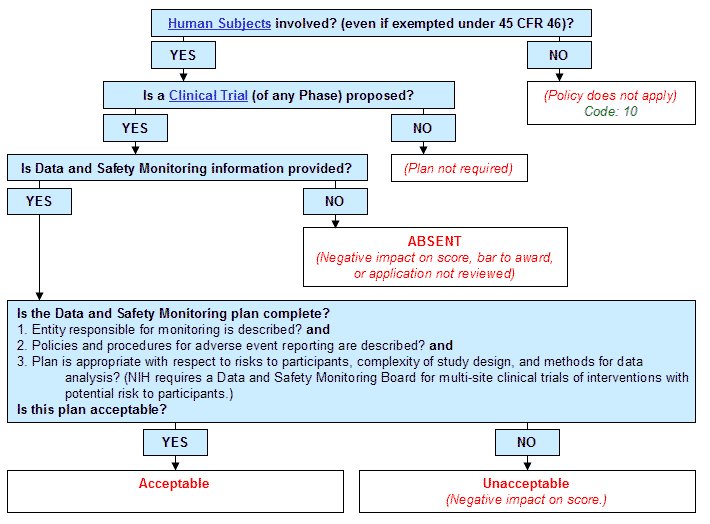
Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

Reasons for frequency of on-site monitoring visits. CTU could choose... | Download Scientific Diagram

Figure 3 from Validation of a risk-assessment scale and a risk-adapted monitoring plan for academic clinical research studies--the Pre-Optimon study. | Semantic Scholar
PLOS ONE: Pilot, double-blind, randomized, placebo-controlled clinical trial of the supplement food Nyaditum resae® in adults with or without latent TB infection: Safety and immunogenicity
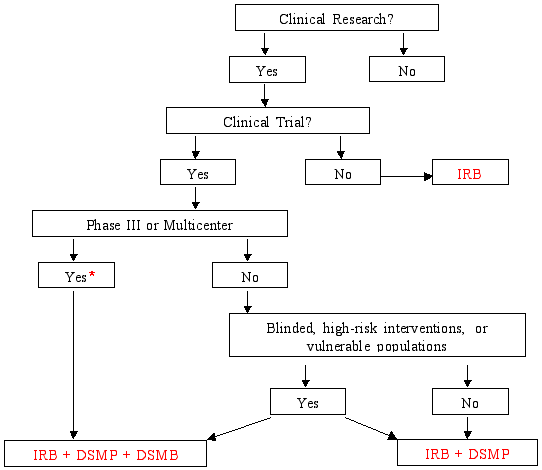
Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)